5 June 2025
Categories: 1-Minute Read, News, Press ReleasesActivInsights is proud to announce the opening of its first U.S. office, in Texas.
7 April 2025
Categories: 1-Minute Read, Insight, NewsA global effort to prioritise the protection of women’s long-term health and well-being is fundamental
13 March 2025
Categories: 1-Minute Read, Insight, NewsSleep is the most important of our 24-hour physical behaviours. Measure sleep in real-world conditions.
13 November 2024
Categories: 1-Minute Read, Insight, NewsActivinsights presents the digital measures advancements in clinical trials at summit.
15 January 2024
Categories: 1-Minute Read, News, Press ReleasesWe are delighted to share the exciting news that Activinsights
5 December 2022
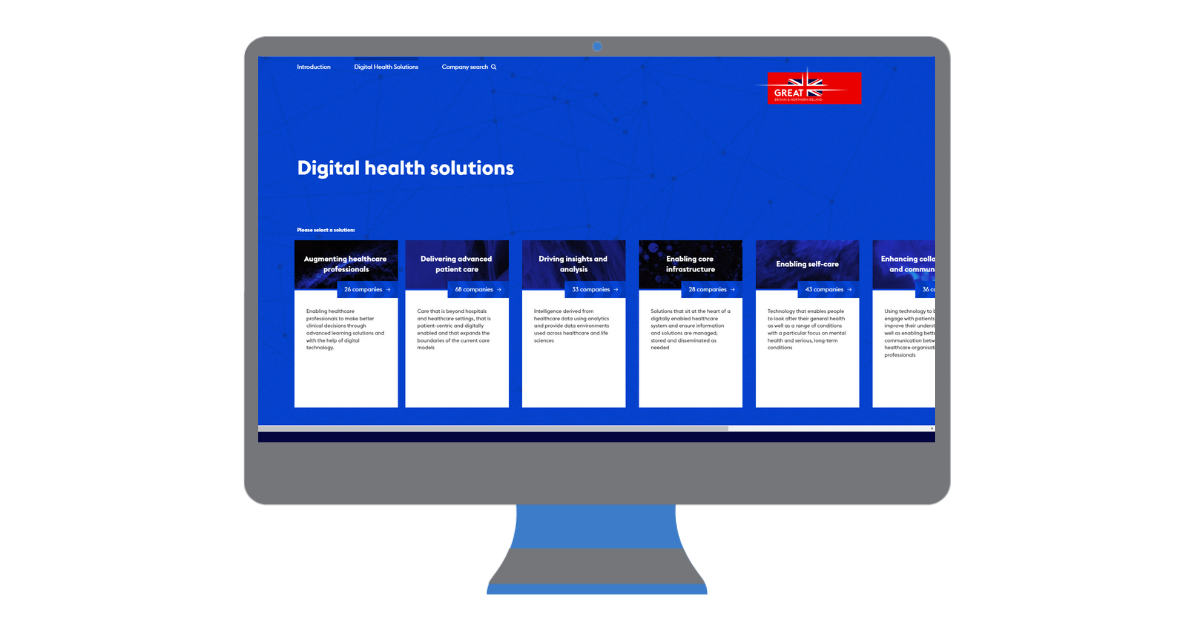
Categories: 1-Minute Read, Insight, NewsActivinsights Feature in the Department for International Trade's 2023 UK

Contact Us
6 Nene Road
Bicton Industrial Park
Kimbolton
Cambs
PE28 0LF
info@activinsights.com
+44 (0)1480 862082

Contact Us
6 Nene Road
Bicton Industrial Park
Kimbolton
Cambs
PE28 0LF
info@activinsights.com
+44 (0)1480 862082

Contact Us
6 Nene Road
Bicton Industrial Park
Kimbolton
Cambs
PE28 0LF
info@activinsights.com
+44 (0)1480 862082
Site Map
Digital Health Technologies
Public Health Research
Clinical Trials
Study Services
Expertise
Company
Resources